Efficient synthesis of chiral compounds under mild conditions is highly desirable in the chemical and pharmaceutical communities, but it often faces difficulties. Although various enzymes have been harnessed as biocatalysts in asymmetric reduction, identifying key residues that function as stereoselectivity-switches constitutes a major challenge.
For this purpose, the SUN Zhoutong group from Tianjin Institute of Industrial Biology, Chinese Academy of Sciences address this question by using the Zn-dependent alcohol dehydrogenase from Thermoanaerobacter brockii (TbSADH) as a model enzyme in the asymmetric reduction of a difficult-to-reduce ketone. The analysis of epistatic interactions was performed in order to determine the key residues that influence the respective selectivity. Furthermore, molecular dynamics (MD) simulations as well as quantum mechanical (QM) calculations were applied to the respective single, double and triple mutants and related variants, thereby shedding light on the factors that determine the inversion of stereoselectivity at the atomic and molecular level.
This research has been published online in Catalysis Science & Technology, entitled “Unraveling the mechanism of enantio-controlling switches of an alcohol dehydrogenase toward sterically small ketone”.
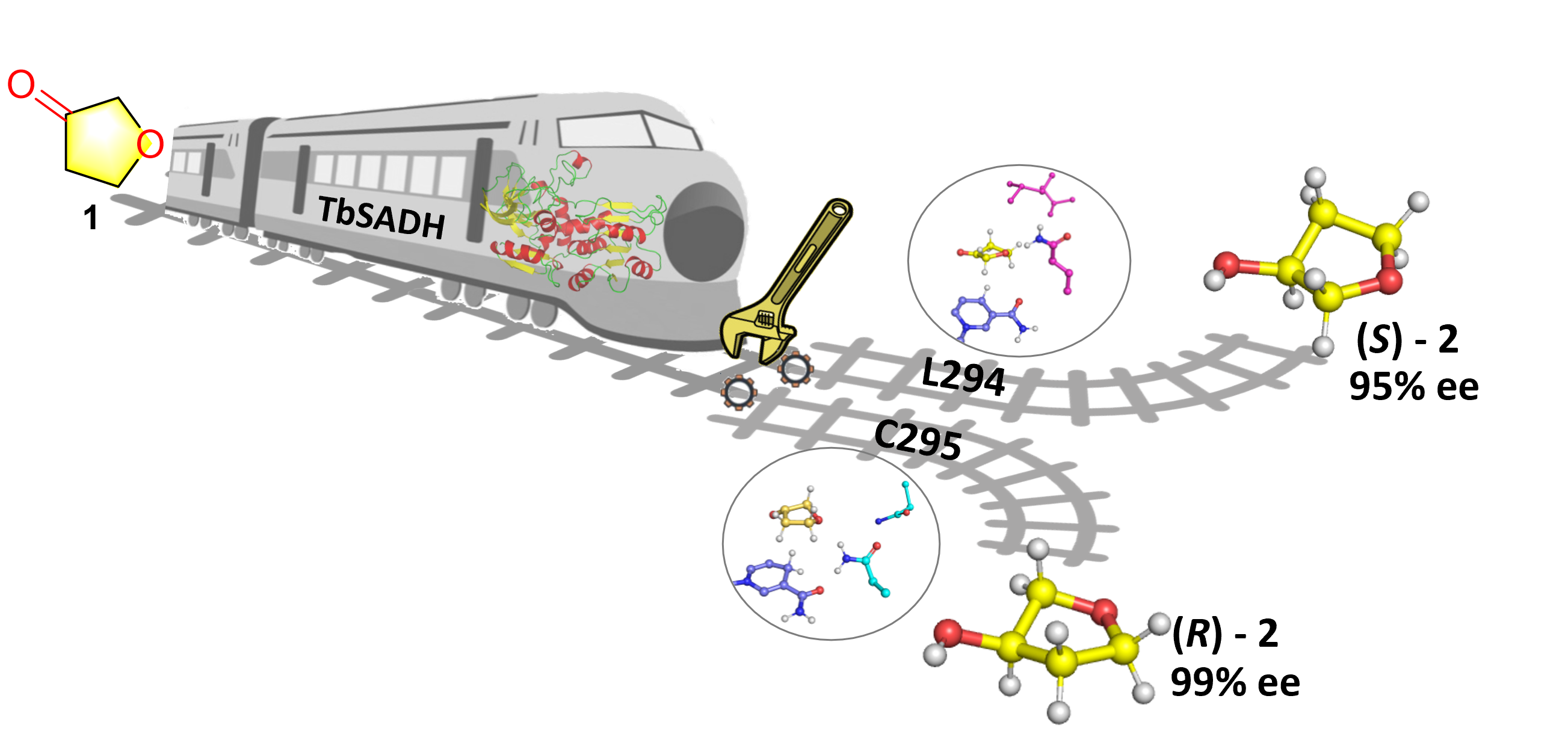
Scheme. Unraveling the Mechanism of “Switches” that specifically control the inversion of product chirality. ( Image by TIBCAS )
Contact
Prof. SUN Zhoutong
Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences.
Phone: (0)22–84861981
Email: sunzht@tib.cas.cn