The Kolbe-Schmitt reaction is a carboxylation reaction proceeded by heating sodium phenolate (the sodium salt of phenol) and carbon dioxide in alkaline under pressure (100 atm, 125 °C) to produce salicylic acid, which is one of the most important ways of industrial salicylic acid synthesis and its derivatives. Salicylic acid and its derivatives are widely used in the synthesis of dyes and pharmaceutical products, such as aspirin, insecticide and so on.
Aromatic carboxylic acid decarboxylases are used to catalyze the decarboxylation of the carboxylic acids and the reversible carboxylation of aromatics (Kolbe-Schmitt reaction) in aqueous phase under atmospheric pressure, which avoids the severe reaction conditions of traditional Kolbe-Schmitt reaction. However, the conversion rate is lower than 50% even under the optimal condition due to the thermodynamic equilibrium of the reaction.
A team led by Professor ZHU Dunming and Professor WU Qiaqing in National Engineering Laboratory for Industrial Enzymes at Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences (TIB, CAS) has solved the problem of the low conversion in the reversible reaction. Recombinant E. coli containing aromatic carboxylic acid decarboxylase catalyzed the carboxylation. With quaternary ammonium salts being the precipitant in the reaction system, the equilibrium of reaction was driven to the carboxylation, resulting in high yield of carboxylation, which can be as much as 97%. The enzymatic Kolbe-Schmitt reaction provides a new way for carbon dioxide fixation and synthesis of aromatic carboxylic acid.
Being financially supported by the National Natural Science Foundation of China (Grant No. 21472232), the work entitled “An Unprecedented Effective Enzymatic Carboxylation of Phenols” has been published in ACS Catalysis. REN Jie, PhD candidate of TIB, is the first author of this paper.
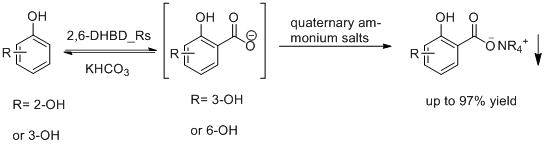
Enzymatic Kolbe-Schmitt reaction (Image by TIB)
Contact:
Pro. WU Qiaqing
Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences
wu_qq@tib.cas.cn